
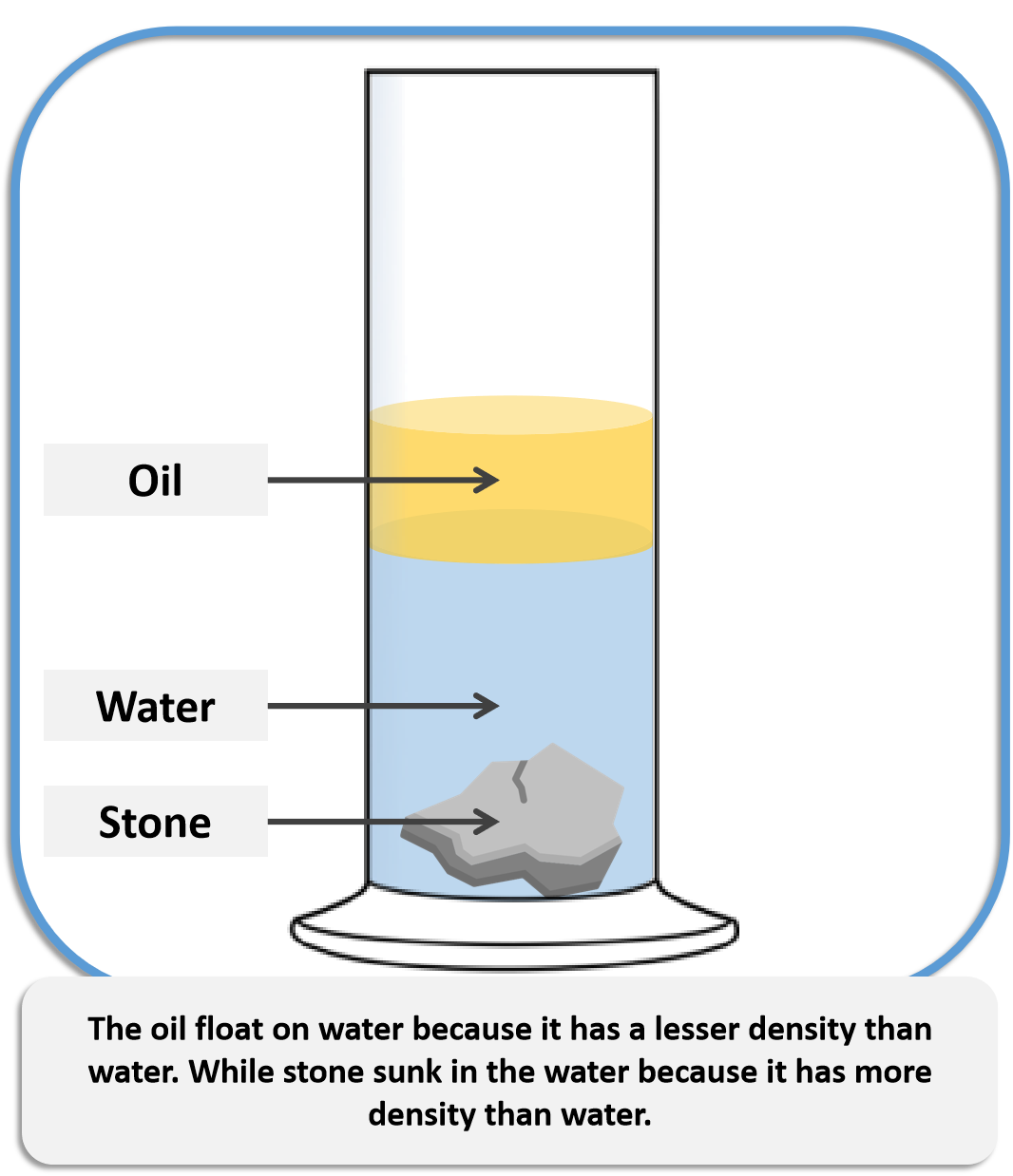
Another application is to determine whether one substance is denser than another by comparing their densities. It is, on the other hand, extremely useful in predicting whether or not something will float when submerged in water. Specific gravity values of a substance are not commonly encountered in everyday life.
When it comes to domestic and commercial applications, density is frequently used however, specific gravity is more commonly encountered when measuring the concentration of solutions in industrial settings.The symbol for density is and is represented in kilograms per cubic metre. Specific gravity, on the other hand, does not have a SI unit and for density it is represented in kilograms per cubic metre.When it comes to density, the absolute value is used, whereas specific gravity is represented in the relative value.
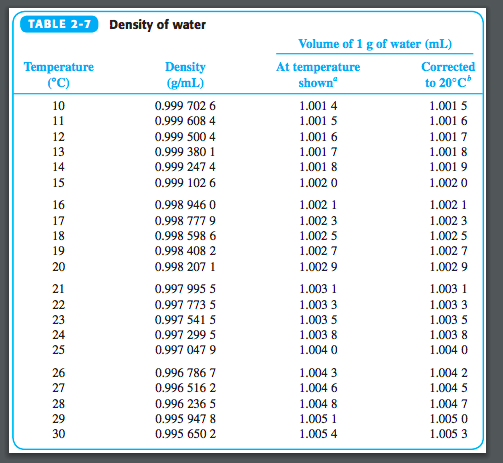
The density of a substance can be calculated by knowing its weight and volume, whereas the specific gravity of a substance can be calculated by dividing the density of a substance by the density of water.Density and specific gravity are two different concepts. Density is defined as the mass of an object per unit volume, whereas relative density, also known as specific gravity, is defined as the ratio of a substance’s weight to the weight of the same volume of water.Difference Between Density and Specific Gravity Therefore, any change in temperature or pressure will have an effect on the density of a substance. Specific gravity is defined as the ratio of the density of a substance or liquid to the density of water at a given temperature and pressure under certain conditions. It is common practice to use the density of water at 4oC as a reference point because water at this temperature has the highest density of 1000 kg/m3, making it an excellent reference point. Here, ρsubstsnce = Density of the substance being measured When a substance has a density greater than or equal to that of water at a specified temperature, it is said to have Specific gravity, which is a dimensionless quantity defined as the ratio of the density of the substance to the density of water at that temperature and is expressed as Increasing the heat convection from the bottom to the top of a fluid in most materials occurs due to the decreased density of the heated fluid, which causes it to rise in comparison to the denser unheated material in most materials. With a few exceptions, raising the temperature of a substance causes its density to decrease by increasing the volume of the substance. Increasing the pressure applied to an object causes the object’s volume to shrink, resulting in an increase in its density. Solids and liquids typically exhibit only minor variations, whereas gas exhibits significantly greater variations.

The density of a material changes in response to changes in temperature and pressure. Density is defined mathematically as the product of mass divided by volume: The symbol for density most commonly used is ρ (the lowercase Greek letter rho), although the Latin letter D can also be used to represent the quantity. The density of a substance is defined as the mass per unit volume of the substance. It is the measurement of density in relation to the density of pure water that is referred to as specific gravity, also known as relative density. Density is calculated by dividing the mass by the volume of the object. Density, on the other hand, is measured in terms of weight units in relation to the size of the object. Water is commonly used as a standard of comparison. Specific gravity is calculated by dividing the density of a substance by the density of a standard or reference substance.
